Growing Beef Newsletter
April 2026, Volume 16, Issue 10
Best management practices for spring processing
Chris Clark, ISU Extension and Outreach beef specialist
It will soon be time to process spring-born calves, which brings up the topic of best management practices and following Beef Quality Assurance (BQA) principles for all treatments. The overall concepts are pretty simple, but it takes attention to detail to get the most out of each treatment and to ensure our product is as safe, wholesome and palatable as possible.
Here are ten simple reminders to help you with those details.
- Follow label directions for all treatments including injections, implants, pour-ons, insecticide ear tags, etc.
- Administer all subcutaneous and intramuscular injections in front of the shoulder in the injection site triangle of the neck.
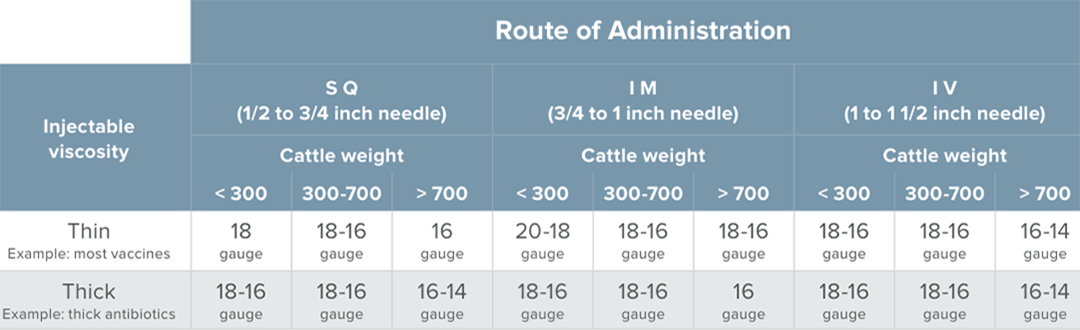
- Select the appropriate needle diameter and length based on the weight of the animals being treated, viscosity of products being injected, and routes of administration. Needles should be small enough to minimize tissue damage but large enough to prevent bending and breaking. The diameter should be appropriate for the viscosity of the product, and the length should be appropriate for the route of administration. For young calves weighing less than 300 pounds, 18 gauge needles are reasonable for most vaccines. For subcutaneous injections, ½ inch to ¾ inch needle length should work well and for intramuscular injections, ¾ inch to 1 inch needle length should be appropriate. Keep in mind the greater the needle gauge the smaller the diameter and vice versa.
*Please refer to the BQA chart below for more information on needle selection. - Practice injection technique and pay attention to angle of injection and the feel of the needle within the tissue. Subcutaneous injections should be applied at approximately 45 degrees to the body and intramuscular injections should be applied at approximately 90 degrees to the body. With experience, you can learn to feel whether you are in that subcutaneous space or whether you have entered the underlying muscle.
- Change needles frequently. At a minimum, needles should be changed every 10-15 head. Additionally, a new needle should always be applied before refilling a syringe and any bent or burred needles should be immediately replaced.
- For reusable syringes, clean well after each use by thoroughly rinsing with hot water. Refrain from using soaps and disinfectants because residues of these substances can damage vaccines and reduce vaccine efficacy.
- Handle vaccines with care. When using modified live vaccines, mix only what you can use in an hour. Keep vaccines at steady, reasonable temperatures and take care to avoid freezing, excessive heat and exposure to UV light. Reconstitute modified live vaccines with sterile transfer needles and roll or invert gently to mix rather than shaking vigorously.
- When possible, choose subcutaneous routes of administration over intramuscular routes. Some products are labeled to be given either way and when you have the choice, choose subcutaneous. Any insertion of a needle or injection of a substance into muscle tissue will cause tissue damage, potentially impacting the quality of that product.
- Document complete processing/treatment records including animal or group identification, treatment date, products administered, withdrawal times, earliest date animals would clear withdrawal times, dose administered, route of administration, name of person administering drugs and any prescription information.
- Do not mix different vaccines or drugs in the same syringe or use a syringe to administer different products without washing in between. Try to place injections at least 4 inches apart from other injections to avoid product mixing/interaction within animal tissue.
For more information about BQA practices, please refer to bqa.org for access to resources and online certification courses. For a list of upcoming in-person BQA trainings in the state of Iowa, please refer to this Iowa BQA page.
Attention to these details can help to maximize vaccine efficacy, protect the safety of our product, and promote consumer confidence and demand. Strive for excellence in processing this spring to promote the health and performance of your cattle and ensure beef quality and safety for consumers.
*Recommended needle size based on animal weight, viscosity of product, and route of administration. Information from the current BQA Field Guide.
This monthly newsletter is free and provides timely information on topics that matter most to Iowa beef producers. You’re welcome to use information and articles from the newsletter - simply credit Iowa Beef Center.
